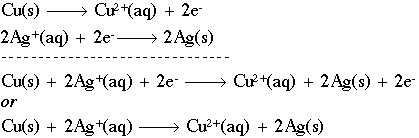
Each of the two partial equations involves the same number of electrons. Multiply each partial equation by a suitable factor for balancing ionization chemicals. These electron particles involve balancing oxidation and reduction half-reactions. Balancing the changes of partial chemical equations by adding a suitable number of electrons.If the reaction occurs in acid solution or low pH solution use a requisite number of hydrogen ions for balancing the number of the atom involved in the partial equations and for the alkaline solution or high pH solution we use hydroxyl ions.Set up the partial balancing equations representing the reduction of the oxidant, and the oxidation of the reductant.Certain the oxidizing agent and reducing agent, and their chemical formula for balancing chemical equations.We use rules for balancing equations by the ion-electron method are Balancing redox reactions by ion electron methodīalancing chemical equations by the ion-electron method has certain general rules or formulas which use for balance oxidation-reduction reactions or redox reactions.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
